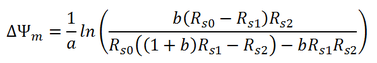An Application: Estimation of Mitochondrial Membrane Potential (MMP) in Culture Bovine Pulmonary Arterial Endothelial Cells
using TMRE or Rhodamine 123
using TMRE or Rhodamine 123
Estimation MMP Using the Temporal Kinetic Model
Download Model Codes
|
Firstly, the binding-unbinding coefficents between TMRE and cuvette was estimated using the data in the absence of cells..
Estimated values using data in above figure:
K1 = 0.005687; [binding constant ] K-1 = 0.038545; [unbinding reacting rate] | |||||||
|
Then, the temporal kinetic model was fitted to above minimum set of experimental data set as described in cell model section.
The estimated parameters in the model are mitochondrial membrane potential, plasma membrane potential, vitrual cellular volume, permeability-surface area product, mitochondrial permeability-surface area product, maximum efflux rate of P-glycoprotein. All of other variables in the model are known. Table 1 below listed the estimated results by fitting the model to mean values of experimental data:
These results are based on model fitting to the mean value of several experimental sets.
However, it is known that variances exist among diffrent experimental sets. These experimental sets could have different initial TMRE concentration in the extracellular medium since the extracellular medium were prepared each time, different cell status since they were different batches. Thus it might be more reasonable to use the individual minimum data sets, which had the same initial TMRE concentration and the same batch of cells, to estimate above parameters instead of the mean values of multiple sets, especially when there was a big variance between data sets. Table 2 below listed the average of results estimated by fitting the model to data sets one by one. |
Discussion: The indepence of this method on probes
To validate the estimated values and the independence of this method to probe, the similiar experiments were carried out using another rhodamine dye - Rhodamine 123 whose membrane permeability is smaller than TMRE and hence need a longer distribution time.
|
Table 3 below listed the estiamted results using the mean values of experimental data:
Table 4. below listed the average of results estimated by fitting the model to minimum data set one by one.
Discussions:
1. The results estimated using R123 data are comparable to the results using TMRE dat, this demonstrate the independence of this method on probes. 2. The results estimated by fitting the model to individual data sets are more consistent than the values estimated by the mean value of data sets, especially for R123 set. This suggests that using the mean values from different data sets might not be a good way to run the model fitting. The abnormal small plasma membrane potential estimated by mean R123 data set is quite possible due to the variance among different R123 data sets such as the different initial R123 concentration. So it is highly recommended to use individual data set to run model fitting. 3. The model fitting didn't give a good estimation for P2S2 which may be due to the insufficient information from the experimental data. A possible explanation is that TMRE/R123 was driven into mitochondria mainly by mitochondrial membrane potential and the effect of concentration gradient across mitochondrial membrane is neglectable. Therefore, there are no sufficient information in the experimental data to estimate P2S2. Since P2S2 didn't play a big role on driving TMRE/R123 into mitochondria, the variance in P2S2 value has no significant effect on TMRE/R123 distribution. This point was demonstrated in our model simulation using different P2S2. |
Discussion: The Model Validation and The Effect of the inhibitor CCCP
To validate the model, the estimated parameters are used to predict the experimental data which were not used for model fitting. Since the data set using TMRE have a very good consistency, where the estimated parameters using the mean data and the average of parameters estimated using individual data sets are quite close, we used the mean TMRE experimental data for data validation.
|
Figure: The predicted results using the estimated parameters listed in Table 1.
The assumptions for these predictions are the effect of inhibitors, which are, 1) The proton pore CCCP is assumed to dissipate mitochondrial membrane potential completely. 2) High potassium (HK) media dissipate plasma membrane potential completely. 3) GF inhibits multi-drug pump completely. |
Frankly speaking, these predictions are miserable. Something was wrong! The mistake would cause by the model or by the assumptions.
By observing the data, it is noticable that TMRE concentrations under steady state with CCCP only and with CCCP and HK are very close. Itsuggests that CCCP might not only dissipate mitochondrial membrane potential but also plasma membrane potential. This guess was also supported by some previous publications such as Daniel L. Farkas' paper. (Biophys J. 1989 Dec;56(6):1053-69) where it showed that FCCP partially dissipated plasma membrane potential by imaging technique. So, we udpated the assumption for the effect of CCCP as below: CCCP dissipates mitochondrial membrane potential completely and dissipates plasma membrane potential partially. The dissipation of plasma membrane potential follows Gaussian equation with a delay t. There is no special reason to make Gaussian equation for this process. Since the process how CCCP dissipates plasma membrane potential is unknown, so I just took the most common equation for the natural process.
Fortunately, the parameters we estimated didn't use the experimental data in the presence of CCCP without high potassium, the improper assumption of CCCP's inhibitory effect has no effect on the estimated parameters. So by fitting the data in the presence of CCCP only to the updated model with 2 variables, a, the percentage of dissipated plasma membrane potential and t, the delayed time, we got that a=0.75 and t = 2.3 minutes.
By observing the data, it is noticable that TMRE concentrations under steady state with CCCP only and with CCCP and HK are very close. Itsuggests that CCCP might not only dissipate mitochondrial membrane potential but also plasma membrane potential. This guess was also supported by some previous publications such as Daniel L. Farkas' paper. (Biophys J. 1989 Dec;56(6):1053-69) where it showed that FCCP partially dissipated plasma membrane potential by imaging technique. So, we udpated the assumption for the effect of CCCP as below: CCCP dissipates mitochondrial membrane potential completely and dissipates plasma membrane potential partially. The dissipation of plasma membrane potential follows Gaussian equation with a delay t. There is no special reason to make Gaussian equation for this process. Since the process how CCCP dissipates plasma membrane potential is unknown, so I just took the most common equation for the natural process.
Fortunately, the parameters we estimated didn't use the experimental data in the presence of CCCP without high potassium, the improper assumption of CCCP's inhibitory effect has no effect on the estimated parameters. So by fitting the data in the presence of CCCP only to the updated model with 2 variables, a, the percentage of dissipated plasma membrane potential and t, the delayed time, we got that a=0.75 and t = 2.3 minutes.
Then we predicted the results in the presence of CCCP+GF with the estimated variables and the updated model as showed below.
These two values were validated by R123 data lso asince the effect of CCCP should not affect by the selected probe.
These two values were validated by R123 data lso asince the effect of CCCP should not affect by the selected probe.
Estimating MMP using a Steady State Model.
Steady State Model
It is noticable that in the presence of both GF120918 and high potassium in extracellular medium, mitochondrial membrane potential would be the only factor affecting TMRE distribution under steady state.
In the presence of GF120918, high potassium and CCCP simutaneously, then TMRE would distribute evenly under steady state, which allows us to estimate the volume of cells.
This will make it possible to estimate mitochondrial membrane potential using a set of [TMREe] values under steady state.
While, the total mitochondrial volume was needed to estimate the contribution of mitochondrial membrane potential to TMRE distribution. In this study, the total volume of mitochondria was estimated based on the published studies, which was 2% of total volume of endothelial cells. This ratio could vary according to the type of cells.
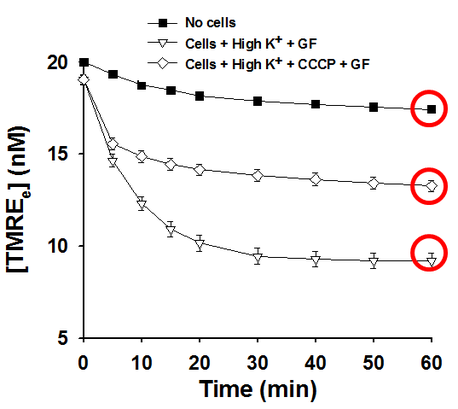
Though theoretically it takes infinite time to reach steady state of TMRE distribution, it is possible to reach the state close enough to steady state in a limited time as showed in figure below.
In the presence of GF120918, high potassium and CCCP simutaneously, then TMRE would distribute evenly under steady state, which allows us to estimate the volume of cells.
This will make it possible to estimate mitochondrial membrane potential using a set of [TMREe] values under steady state.
While, the total mitochondrial volume was needed to estimate the contribution of mitochondrial membrane potential to TMRE distribution. In this study, the total volume of mitochondria was estimated based on the published studies, which was 2% of total volume of endothelial cells. This ratio could vary according to the type of cells.
Though theoretically it takes infinite time to reach steady state of TMRE distribution, it is possible to reach the state close enough to steady state in a limited time as showed in figure below.
|
Mitochondrial membrane potential could be estimated by the equation below: .
a: constant, a=ZF/RT=0.0374 /mV
b: ratio between total mitochondrial volume and total cell volume. For endothelial cells, b=0.02 Rs0: Extracellular TMRE concentration under steady state in the absence of cells (the value in the top red circle) Rs1: Extracelllular TMRE concentration under steady state in the presence of cells, GF120918, CCCP and high potassium ( the value in the middle red circle) Rs2: Extracellular TMRE concentration under steady state in the presence of cells, GF120918 and high potassium. ( the value in the bottom red circle) | ||||||
Related Publication:
Quantifying mitochondrial and plasma membrane potentials in intact pulmonary arterial endothelial cells based on extracellular disposition of rhodamine dyes
Gan Z, Audi SH, Bongard RD, Gauthier KM, Merker MP.
Am J Physiol Lung Cell Mol Physiol. 2011 May;300(5):L762-72
Quantifying mitochondrial and plasma membrane potentials in intact pulmonary arterial endothelial cells based on extracellular disposition of rhodamine dyes
Gan Z, Audi SH, Bongard RD, Gauthier KM, Merker MP.
Am J Physiol Lung Cell Mol Physiol. 2011 May;300(5):L762-72